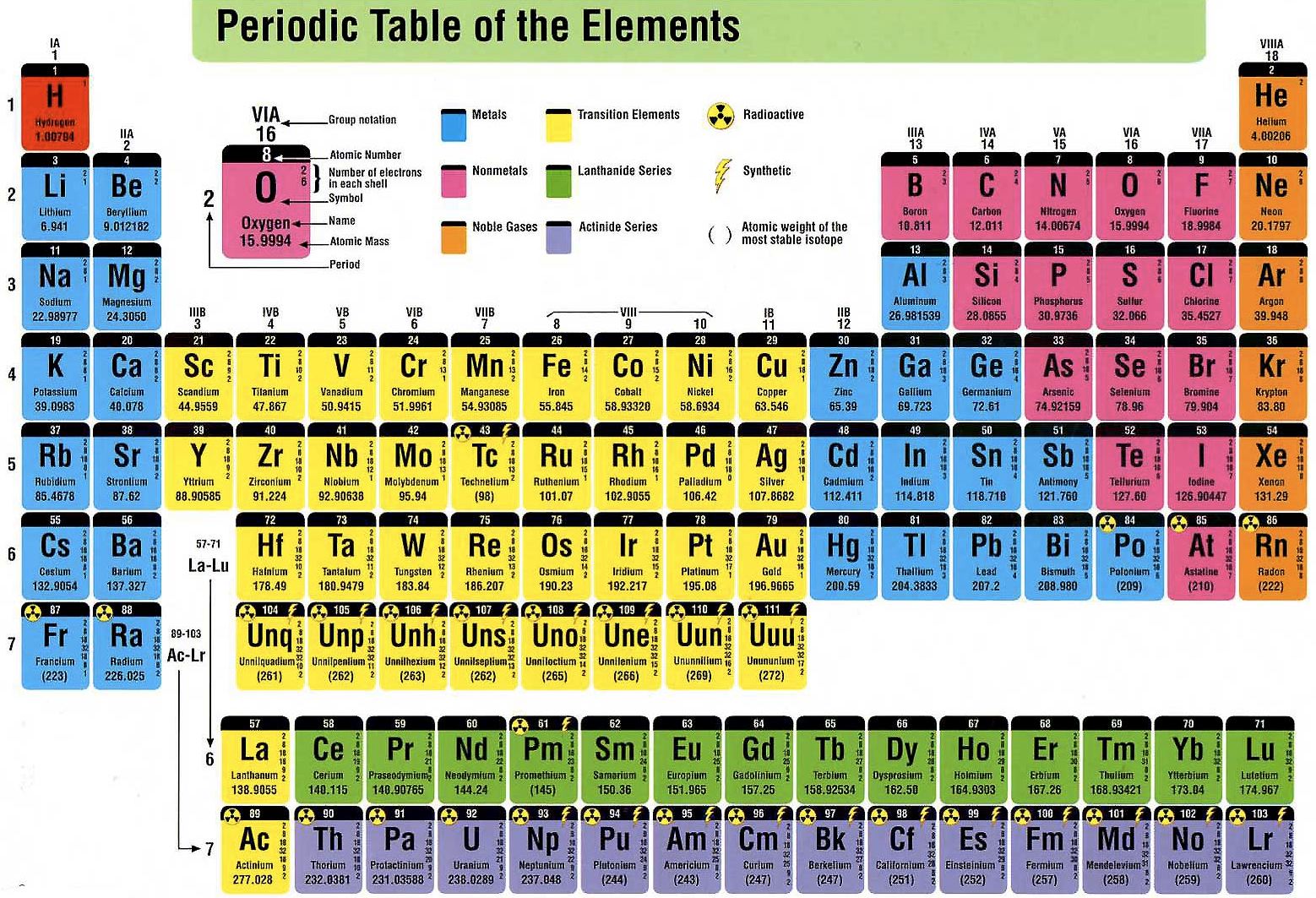
Phosphorus is an integral part of ATP, the energy currency of our body.Ĭalcium (Ca)- Calcium helps in maintaining bone strength. Phosphorus (P)- Phosphorus is used in the military to make weapons (white phosphorus). Silicon (Si) - Silicon, a semiconductor, is used in computer chips. also contain carbon.Īluminium (Al)- Aluminium, being malleable and soft, is used in the making of various products like utensils, aeroplane parts, window frames etc. Protein, sugar and other essential compounds including glucose contain carbon. Oxygen is generated during the process of photosynthesis in plants and different types of microorganisms.Ĭarbon (C)- Carbon consists of 18% of the human body. Humans, along with many other beings, require oxygen for breathing. Respiration is the energy-producing mechanism that dictates the metabolisms of most living organisms. Oxygen (O)- Oxygen has an essential role in respiration. Some of the elements of the first 20 elements of the periodic table are very much helpful for our daily life such as: How are the first 20 elements of the periodic table useful for us? As we know that the atomic number is equal to the number of electrons, we can easily predict the atom's electronic configuration by merely knowing its atomic number. 
It has 11 electrons surrounding the nucleus. Similarly, in sodium, which has an atomic number of 11, the sodium atom's nucleus consists of 11 protons. This implies that in the neutral state, the number of protons in the nucleus is 8. Let's try to understand it further with the example of Oxygen. It is numerically equal to the number of electrons in a neutral atom. Hence, it is also similar to the number of protons in the nucleus. In other words, we can say that the atomic number is equal to the charge on the nucleus. The number of electrons surrounding the nucleus in a neutral atom. Gives an idea about the number of protons in the nucleus of an atom The atomic number of an element in the periodic table provides us with the following information: The mass of an electron is almost 1/1836 times the mass of a proton. Electrons have an equal charge as the proton, but with a negative sign. The size of the electrons is smaller than that of the nucleus. Electrons tend to orbit around the nucleus. Neutrons, along with the protons, add up to form the nucleus of the atom.Įlectrons - Electrons are also subatomic. A neutron weighs a little bit higher than a proton. Neutrons - It is also a subatomic particle. It resides in the nucleus of an atom of the element. Protons - It is a subatomic (occurring within an atom) particle. Importance of The Atomic Number of An Element in A Periodic Tableīefore knowing the importance of an atomic number of elements take a look at the following definitions.Ītomic number - The atomic number tells us how many protons are there in the nucleus. We created a timeline of the history of the periodic table.Each element consists of distinct properties owing to its atomic number, atomic mass, electronic configuration, electronegativity, electron gain enthalpy, etc. The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. Who invented the Periodic Table of Elements? The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. The Periodic Table of Elements can be used as an assisting tool in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. How is the Periodic Table of Elements used? The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. The Periodic table of elements is a tool, developed by scientists over hundreds of years. List of all the elements and their properties:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
